
These non-polar solvents are usually hydrocarbons that dissolve other organic hydrophobic substances. Most lipids are soluble in non-polar solvents. When amphipathic lipids come in contact with whatever their hydrophilic parts stay in contact with water and the hydrophobic parts stay away from water. Such lipids are usually those which make cellular membranes because of the way they behave in water. Therefore, such lipid molecules are called amphipathic. But this is usually a small part of the molecule, the rest of the molecule is usually hydrophobic. Some lipids show a small tendency of being hydrophilic. However the majority of lipids are hydrophobic, that is, they do not interact with water. 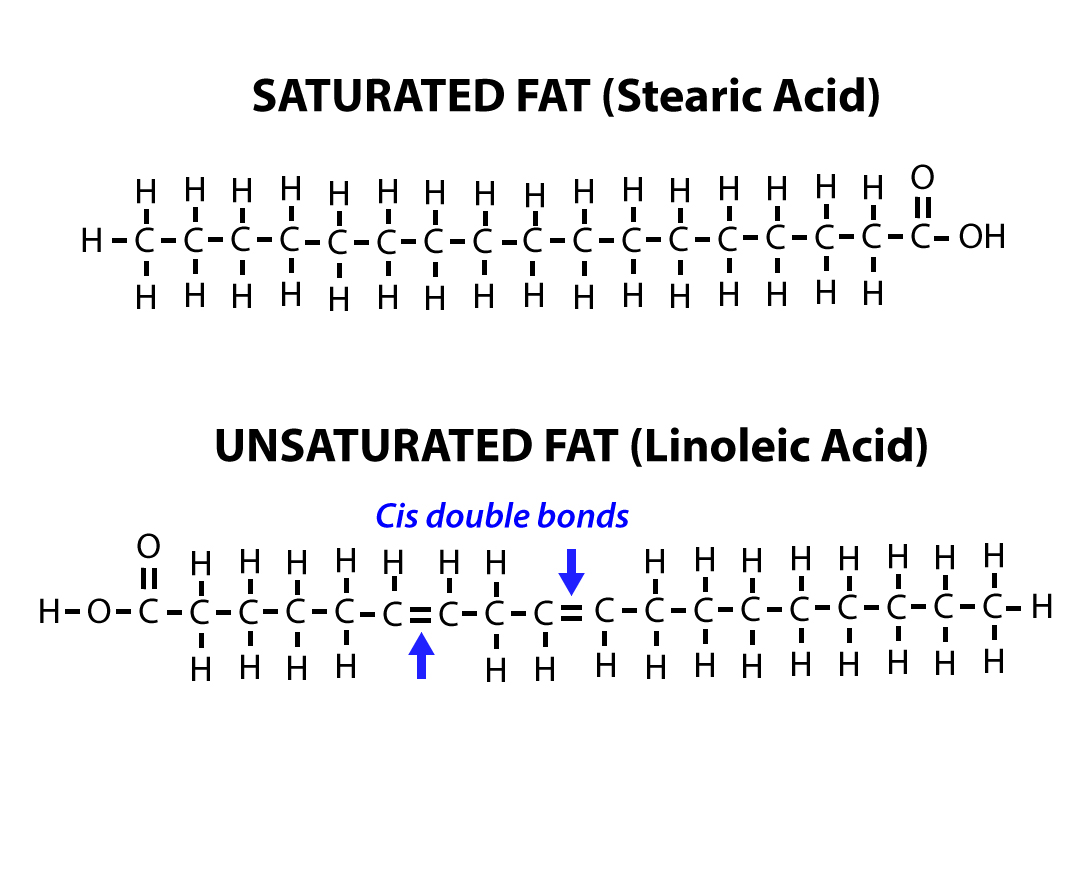
Most biological substances can interact with water. The study of the function and structure of lipids constitutes a sizable part of the study of organic compounds because they make up such substances as oils, fats, wax, membranes and energy-storing molecules in plants and animals. Put in simple terms, most substances made of lipids will not be miscible with water. Lipids are classified as those organic compounds which do not interact with water.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
